IVF is about taking eggs and sperm, putting them together in a lab and letting the natural process happen. If they fertilise, an embryo will form. The tiny embryo (0.1 mm) is then inserted into the uterus. If it grows and develops, you’ll take your baby home 9 months later.
Will I Need IVF?
Your fertility specialist gets to know your medical history and your individual circumstances. There’s lots of reasons we may recommend IVF, and create the best treatment plan for your body.
Some common reasons why IVF may be required include:
- Blocked fallopian tubes
- Fallopian tube damage/tubal factor/tubal ligation
- Male infertility relating to sperm quality or quantity
- Endometriosis
- Uterine fibroids
- Polycystic ovarian syndrome (PCOS)
- Increased difficulty of conceiving naturally as women age
- Unexplained infertility
- Recurring miscarriage
- Potential genetic issues where embryo screening may help.
The IVF Process
An IVF ‘cycle’ is how we describe one round of IVF treatment starting with the first day of your period.
As part of your fertility plan, you may start medication or injections before the first day of the cycle.
Step 1: Day 1 of your Period
The first official day of your IVF treatment cycle is day 1 of your period. Everyone’s body is different, and you will be helped to understand how to identify day 1.
Step 2: Stimulating your Ovaries
The stimulation phase starts from day 1. In a natural monthly cycle, your ovaries normally produce 1 egg. You’ll take medication for 8-14 days to encourage the follicles in your ovaries (where the eggs live) to produce more eggs.
Your specialist prescribes medication specific to your body and treatment plan. It’s usually in the form of injections, which can vary from 1-2 for the cycle, or 1-2 per day. It can be daunting, but you will be shown exactly how and where to give the injections. You can get your partner involved too and watch and learn together to get it right. It quickly becomes a habit and you’ll be an expert in no time.
The most common hormones in the medications used to stimulate the follicles are:
Follicle-stimulating hormone (FSH)
Iuteinizing hormone (LH).
Step 3: Egg Retrieval
Egg retrieval, or egg ‘pick up’, is a hospital day procedure where the eggs are collected from your ovaries. An anaesthetist will get you ready for a general anaesthetic. You’ll be asleep and the procedure takes about 20-30 minutes.
Recovery takes about 30 minutes and you’ll be able to walk out on your own. It’s a good idea to have a support person with you as you won’t be able to drive after the procedure.
Step 4: The Sperm
If you’re a couple planning on using fresh sperm, the male will produce a sample the morning of the egg retrieval. If you are using frozen or donor sperm, our scientists will have it ready in the lab. The sperm is graded using 4 different levels of quality. It’s washed in a special mixture to slow it down so our scientists can spot the best ones under the microscope. A perfect, healthy sperm is not too fat or thin, with a tail that’s not too long or short. The best sperm are selected, and they’re ready and waiting in the lab to be introduced to the eggs.
Step 5: Fertilisation
It’s important the eggs are fertilised quickly. The eggs and some sperm are placed in a dish. They have the chance to find each other and fertilise like they would naturally within your body.
Step 6: Embryo Development
If the sperm fertilises the egg, it becomes an embryo. We put the embryo into a special incubator where the conditions for growth and development are perfect. We create the perfect growing conditions using a mix of amino acids, just like your body would use to nurture the embryo.
Unfortunately, not all eggs will fertilise and reach the embryo stage. The eggs might not be mature or the sperm not be strong enough. We know you’ll be waiting on news, so we’ll keep you up to date with the progress of your egg, sperm and embryo development.
Stage 7: Embryo transfer
If your embryo develops in the lab, you’re ready for it to be transferred into your uterus.The embryo transfer is a very simple process, like a pap smear. It takes about 5 minutes, you’ll be awake, there’s no anaesthetic, and you can get up straight away. You can continue with your day, the embryo can’t fall out if you stand up or go to the toilet.
Step 8: The final blood test
Approximately two weeks after your embryo transfer, you’ll have a blood test to measure your levels of the hormone hCG (human chorionic gonadotropin). HCG in your bloodstream usually means a positive pregnancy test. Your nurse will let you know exactly when you need to have your blood test, as it may vary for some patients.
Intracytoplasmic Sperm Injection (ICSI) is a technique where a single sperm is given a helping hand to ‘enter’ the egg.Since it only takes one single sperm to fertilise the egg, the embryologist catches a single sperm and injects it directly into the centre of the egg. This method is far more successful when there is male infertility problems.
Who can use ICSI?
ICSI can be an option for patients who have:
- An abnormally low sperm count or poor motility
- A high percentage of abnormal sperm or few healthy sperm
- Sperm obtained via testicular biopsy or micro TESE
- Previously low fertilization rates with standard IVF
ICSI can be used with fresh or frozen/thawed sperm. We will choose the best sperm from the sample, based on it being a normal shape, size and motility (movement).
Sperm Selection Process
The healthiest sperm cells tend to have a certain shape and size, particularly an oval head and a long tail which they use to push themselves along as they swim. Infertile men often make fewer such sperm so that sperm selection for ICSI is important.
Next consideration is sperm motility – a sperm cell’s motility is its ability to move itself around and penetrate an egg. This depends on the length and size of its tail. Tails that are curly or doubled up aren’t as efficient when it comes to swimming.
Routinely in ICSI, a small amount of washed and prepared sperm is placed into thick viscous media that slows the sperm down so that they can be selected according to their shape, motility and trajectory. The most “normal” looking and vigorous sperm are selected and then immobilized by squashing their tails with a glass injection needle. One these sperm are sucked into the needle tail-first ready to be injected.
An embryo transfer is the last part of the in vitro fertilization (IVF) process.
During IVF, fertility medications are used to stimulate the ovaries into releasing healthy eggs.

Image Source: Link
These eggs are then removed from a woman’s ovaries and fertilized in a lab. Once the fertilized eggs have multiplied, the embryos are transferred to the woman’s uterus.
For a pregnancy to begin, the embryo must then attach itself to the wall of her womb or uterus.
Under ultrasound guidance with 3-D/4-D real-time technology (the most precise way to transfer embryos), the embryos are instilled into the uterus via a thin, flexible plastic tube, which is gently passed through the opening in the cervix leading to the interior of the uterus. You literally get to watch the embryo transfer into your uterus as it happens! In about 12 days, a blood test for pregnancy is performed to determine whether the transfer resulted in a pregnancy.
Sperm Retrieval Procedures
In some individuals, spermatozoa may not be present in the ejaculate. This condition is called Azoospermia. This can be either due to problems in sperm production itself or due to obstruction to the flow of semen during ejaculation. Reproductive tract obstruction can be acquired – as a result of infection, trauma, an iatrogenic injury which can occur during bladder neck, pelvic, abdominal or inguinoscrotal surgery.
Congenital anomalies may be relatively uncommon in the general population but can occur in up to 2 percent of infertile men. The best-known condition is the congenital bilateral absence of the vas deferens (CBAVD) which occurs in almost all men with cystic fibrosis.
Two techniques – Epididymal sperm retrieval & micromanipulation have revolutionized the treatment of male infertility in the past decade. Men with congenital bilateral absence of the vas defences (CBAVD) or reproductive tract obstruction are now able to achieve pregnancies with the use of these advanced techniques.
MESA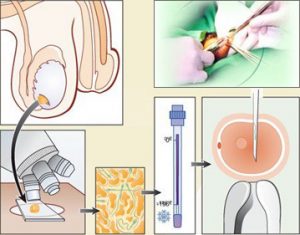
Microsurgical Epididymal Sperm Aspiration (MESA) is used in conditions like obstructive azoospermia, involves dissection of the epididymis under the operating microscope and incision of a single tubule. Fluid spills from the Epididymal tubule and pools in the Epididymal bed. This pooled fluid is then aspirated. Because the epididymis is richly vascularized, this technique invariably leads to contamination by blood cells that may affect sperm fertilizing capacity in vitro.
TESA
TESE or testicular sperm extraction is a surgical biopsy of the testis whereas TESA or testicular sperm aspiration is performed by inserting a needle in the testis and aspirating fluid and tissue with negative pressure. The aspirated tissue is then processed in the embryology laboratory and the sperm cells extracted are used for ICSI.
At Our Clinic, about every third IVF treatment involves an extended blastocyst culture, i.e. the fresh transfer of an embryo at the blastocyst stage. In all our IVF treatments, we strive to choose for one treatment embryos of different ages to be frozen, i.e. both embryos in the cell division stage and blastocysts. This allows us flexibility in planning the time for the frozen embryo transfer.
With blastocyst transfer, embryos are cultured in the laboratory to the blastocyst stage. Fresh embryo transfer is performed in this type of culture 4-5 days after egg collection, instead of the traditional 2-3 days. Blastocysts are frozen when they are four, five or six days old. Blastocyst cultures require optimal laboratory conditions, a culture solution that best supports the development of blastocysts and a low-oxygen culture environment. On average 40% of fertilized eggs reach the blastocyst stage.
is performed in this type of culture 4-5 days after egg collection, instead of the traditional 2-3 days. Blastocysts are frozen when they are four, five or six days old. Blastocyst cultures require optimal laboratory conditions, a culture solution that best supports the development of blastocysts and a low-oxygen culture environment. On average 40% of fertilized eggs reach the blastocyst stage.
The advantage of blastocyst cultures is the efficiency of embryo selection because only the embryos with the best development capacity reach the blastocyst stage. There is a higher probability of pregnancy when using a blastocyst than with an embryo in the cell division stage, with fresh embryo transfer and frozen embryo transfer being the most reliable methods. Blastocysts are also used with excellent results in embryo diagnosis. Blastocyst cultures are particularly useful when predicting a good yield of about 10 eggs. With low egg counts, it poses the risk that no embryo can be transferred if the development of the embryo stops before it reaches the blastocyst stage
Let’s Check Our Math
- 1 in 4 eggs harvested will likely develop to the blastocyst stage.
- 1 in 2 blastocysts will likely have a normal result from preimplantation genetic testing (PGT).
- That means about 1 in 8 eggs will be genetically competent to become a baby.
- There is about a 60% chance that a single embryo with genetically normal PGT result will result in the birth of a baby.
A frozen embryo transfer, or FET, is a kind of IVF treatment where a cryopreserved embryo created in a full IVF cycle is thawed and transferred to a woman’s uterus. The cryopreserved embryo may be from a woman’s previous conventional IVF cycle, or it may be a donor embryo.
Egg donors are resources for women with no eggs due to surgery, chemotherapy, or genetic causes; or with poor egg quality, previously unsuccessful IVF cycles or advanced maternal age. In the egg donor process, eggs are retrieved from a donor’s ovaries, fertilized in the laboratory with the sperm from the recipient’s partner, and the resulting healthy embryos are returned to the recipient’s uterus.
Sperm donation may provide the source for the sperm used in IVF procedures where the male partner produces no sperm or has an inheritable disease, or where the woman being treated has no male partner.
Preimplantation genetic diagnosis(PGD) involves the use of genetic screening mechanisms such as fluorescent in-situ hybridization (FISH) or comparative genomic hybridization (CGH) to help identify genetically abnormal embryos and improve healthy outcomes.


